ACS Omega 2026, 11, 4, 5227-5240 (Image provided)
Author
Kazushige Yokoyama, Kia Haering ’24, Nicole Mathewson ’25, Patrick Loss ’24, and Jani E. Lewis
Publication
Article title
“Visualizing Cytoskeletal Protein Reconstruction of Vulvar Cancer with Surface-Enhanced Raman Spectroscopy and Gold Nanoparticles”
Summary
The research shows that a noninvasive light-based imaging method can identify cells as they shift into a more mobile and aggressive state by detecting changes in their internal structure.
Abstract
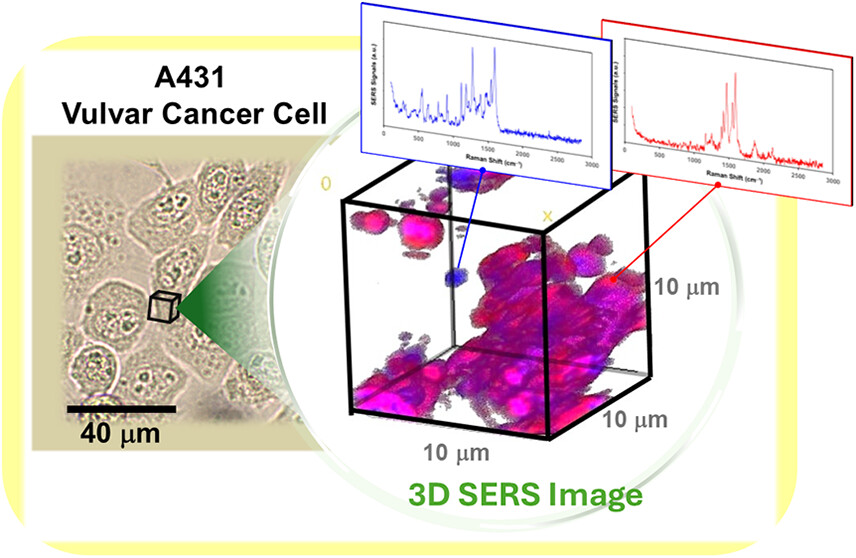
Metastasis of epithelial cancers often involves an epithelial-mesenchymal transition (EMT) characterized by changes in cytoskeletal and adhesion protein expression. This includes loss of the cell adhesion protein, E-cadherin and gain of the cytoskeletal protein, vimentin. Our lab found that the epithelial vulvar cancer cell line A431 undergoes permanent loss of E-cadherin and gain of vimentin expression when treated with a corticosteroid known as clobetasol (referred to as A431D cells). Clobetasol is commonly used to treat chronic vulvar rash making our findings significant when considering repercussions of this treatment. Interestingly, the cells continue to express cytokeratin 8/18 in addition to gaining vimentin expression. The use of Raman spectroscopy has been employed to monitor EMT in breast and oral cancer. We used a 3-dimensional Surface Enhanced Raman Scattering (SERS) imaging by utilizing colloidal gold nanoparticles. By tracking the protein interactions and surface composition in cells before and after treatment with clobetasol the distribution of the transition can be reasoned and visualized. Spectral assignments revealed the order of protein gains and losses while the distinctness of collected signals from literature signals provided information about folding, binding, and repelling forces between these cytoskeletal proteins. The three-dimensional Raman imaging around the cytoplasmic region of the A431D cells exhibited unique features that combined the cytoarchitecture of A431 cells (parent cells expressing cytokeratins 8 and 18) and NIH 3T3 cells (used because they only express vimentin). This suggested Raman imaging could be used to delineate cells which contain multiple types of intermediate filaments and serve as another method to identify cells undergoing an EMT.
Primary research questions
- Can a light-based imaging technique detect when cancer cells shift into a more mobile and aggressive state?
- Does treatment with a commonly used corticosteroid trigger lasting structural and behavioral changes in vulvar cancer cells?
- Can this imaging method distinguish cells that contain multiple types of internal structural proteins during this transition?
- Can three-dimensional imaging reveal how protein organization and interactions change as cells undergo this transformation?
What the research builds on
It was already known that cancer cells can undergo a biological shift that makes them more mobile and invasive, and that this process involves changes in key structural and adhesion proteins. Previous studies had also shown that light-based Raman imaging could monitor similar transitions in certain cancer types.
What the research adds to the discussion
The findings introduce a three-dimensional, label-free imaging method that can distinguish cells with mixed structural features during this transition, providing a new tool to study how cancer cells change and potentially respond to treatment.
Novel methodology
Background: Cancer progression often involves a transition in which cells lose adhesion and gain mobility, accompanied by changes in internal structural proteins. Monitoring these changes typically requires labeling or staining techniques.
Methods: This study applied three-dimensional, surface-enhanced Raman imaging using gold nanoparticles to examine vulvar cancer cells before and after treatment with a commonly used corticosteroid. The technique enabled detailed mapping of protein organization within cells without added dyes.
Results: The imaging approach detected distinct structural patterns in treated cells, including cells that displayed mixed internal structural features. The spectral data also revealed the sequence of protein gains and losses and provided insight into how these proteins interact and reorganize during the transition.
Conclusions: The findings demonstrate that three-dimensional Raman imaging offers a powerful, label-free method to identify and characterize cells undergoing this biological shift, providing a new tool for studying cancer progression and treatment effects.
Implications for society
This research provides a new, noninvasive way to monitor how cancer cells change and respond to treatments. By enabling earlier and more precise detection of cellular transitions, it could help improve cancer research, inform safer use of common medications, and ultimately contribute to better patient outcomes.
Implications for research
This research demonstrates that three-dimensional, label-free Raman imaging can reveal detailed structural changes in cancer cells during key biological transitions. Future studies can build on this approach to investigate how different treatments affect cell behavior, explore similar transitions in other cancer types, and develop new imaging-based methods for studying cellular changes in real time.
Implications for policy
The findings highlight that a commonly used corticosteroid can induce lasting changes in cancer cells. This insight could inform medical guidelines and safety recommendations for the use of such treatments, helping clinicians make more informed decisions and shaping best practices for patient care.
Funding
NSF-MRI Grant #2117780
Citation
K. Yokoyama, K. Haering, N. Mathewson, P. Loss, J.E. Lewis, “Visualizing Cytoskeletal Protein Reconstruction of Vulvar Cancer with Surface-Enhanced Raman Spectroscopy and Gold Nanoparticles”, ACS Omega, Vol. 11 pp. 5227-5240 (2026).
More Research Briefs
Call for Research
We invite SUNY Geneseo faculty to submit peer-reviewed research from the last three years to be added to this site.
